Summary of the Haber Bosch process
|
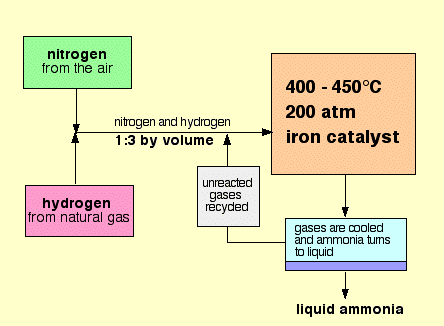
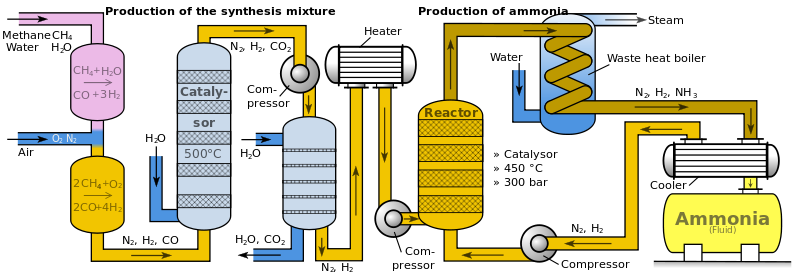
The following flow chart represents the Haber process:
|

History of the developer and the process
He was a German chemist who received the Nobel Prize in Chemistry in 1918 for his development of synthesising ammonia.
Along with Max Born, he developed the Born-Haber Cycle during 20th century. It used to be used to make explosives in the past, but now it has many commercial uses. The cycle helps in the production of ammonia, which is helpful in the production of nitrogen fertilizers. It makes them extremely affordable, and due to this, there has been a population boom due to the increase in world food production. However, nitrogen fertilizers also cause natural bacteria to produce nitrous oxide which is a greenhouse gas and can also cause acid rain.
The development of the process was a result of the World War 1 where Haber used the process to help the German army developing explosives with the help of Carl Bosch. Haber took the particular step when the British Army had cut off the supplies of fertilisers from Chile to Germany. The main aim of his invention at that time was to help the German Army get through the world war. That is why, at times, it is considered that the World War 1 caused the invention of one of the most significant developments in Chemistry - THE HABER PROCESS.
He was a German chemist who received the Nobel Prize in Chemistry in 1918 for his development of synthesising ammonia.
Along with Max Born, he developed the Born-Haber Cycle during 20th century. It used to be used to make explosives in the past, but now it has many commercial uses. The cycle helps in the production of ammonia, which is helpful in the production of nitrogen fertilizers. It makes them extremely affordable, and due to this, there has been a population boom due to the increase in world food production. However, nitrogen fertilizers also cause natural bacteria to produce nitrous oxide which is a greenhouse gas and can also cause acid rain.
The development of the process was a result of the World War 1 where Haber used the process to help the German army developing explosives with the help of Carl Bosch. Haber took the particular step when the British Army had cut off the supplies of fertilisers from Chile to Germany. The main aim of his invention at that time was to help the German Army get through the world war. That is why, at times, it is considered that the World War 1 caused the invention of one of the most significant developments in Chemistry - THE HABER PROCESS.
How the Haber-Bosch process works:
1. Firstly, Nitrogen and Hydrogen (the raw materials), are scrubbed, or cleaned, to remove any impurities.
2. The mixture of Hydrogen and Nitrogen is compressed, until the pressure reaches 200 atmospheres.
3. The compressed gases now flow to the converter. In the converter, beds of iron are kept at 450 degrees celcius. Iron is used as a catalyst, since this is a reversible reaction. Iron forces the reaction to the right, creating ammonia. Only 15 % of the total mixture forms ammonia.
4. The mixture is then forced into a cooler. Ammonia condenses to a liquid, forming at the bottom of the cooler, but the rest of gases are recycled to attempt to form more ammonia.
5. The liquid ammonia is collected.
1. Firstly, Nitrogen and Hydrogen (the raw materials), are scrubbed, or cleaned, to remove any impurities.
2. The mixture of Hydrogen and Nitrogen is compressed, until the pressure reaches 200 atmospheres.
3. The compressed gases now flow to the converter. In the converter, beds of iron are kept at 450 degrees celcius. Iron is used as a catalyst, since this is a reversible reaction. Iron forces the reaction to the right, creating ammonia. Only 15 % of the total mixture forms ammonia.
4. The mixture is then forced into a cooler. Ammonia condenses to a liquid, forming at the bottom of the cooler, but the rest of gases are recycled to attempt to form more ammonia.
5. The liquid ammonia is collected.
The Haber Process is a good example of the use of equilibrium principles. The equation for the reversible reaction is :
N2(g) + 3H2(g) <--> 2NH3(g) + 92 kJ
Because raising the temperature will increase the speed of both the forward and reverse reactions, a high temperature should bring the reaction to equilibrium rapidly. However, raising the temperature favors the endothermic reaction, shifting the equilibrium in this case to the left, lowering the yield of ammonia, and increasing the time needed to obtain a given quantity of ammonia.
N2(g) + 3H2(g) <--> 2NH3(g) + 92 kJ
Because raising the temperature will increase the speed of both the forward and reverse reactions, a high temperature should bring the reaction to equilibrium rapidly. However, raising the temperature favors the endothermic reaction, shifting the equilibrium in this case to the left, lowering the yield of ammonia, and increasing the time needed to obtain a given quantity of ammonia.
Sources: http://geography.about.com/od/economic-geography/a/Overview-Of-The-Haber-bosch-Process.htm
http://feedingtheworld.wordpress.com/how-it-works/
http://feedingtheworld.wordpress.com/how-it-works/